Description
Primary hyperoxaluria type 1 is a rare disease in which urine oxalate is too high, which over time can harm the kidneys. Nedosiran is indicated to lower urinary oxalate levels in people nine years of age and older with primary hyperoxaluria type 1 and relatively preserved kidney function.
The most common side effects include injection site reactions.
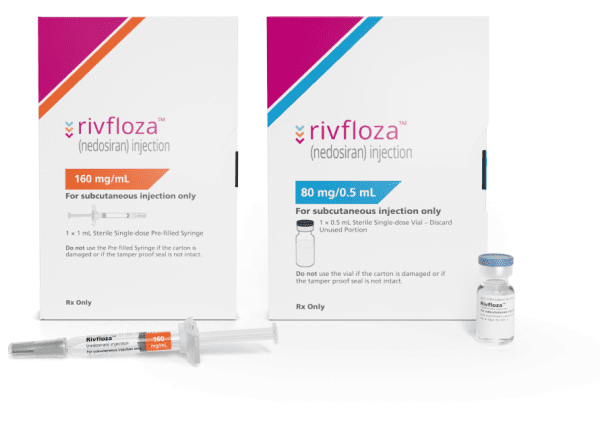
RIVFLOZA® (nedosiran) injection, for subcutaneous use Initial U.S. Approval: 2023
See full prescribing information : Click Here
How to get access RIVFLOZA (nedosiran) for personal use ?
To get access to RIVFLOZA (nedosiran), you need a prescription from a doctor, as it is a prescription-only medication. Your prescription will be filled through a specialty pharmacy, which will handle insurance and coordinate delivery of the medication to you. Your doctor will also need to provide specific information for the prescription to be filled correctly.
However, patients in India can legally access it through the provision called the Named Patient Program (NPP). RIVFLOZA (nedosiran) prices may fluctuate over time because of market dynamics and regulatory changes. It can be imported for personal use under “Named Patient Program” treatment in multiple countries, including the Singapore, Kuala Lumpur (Malaysia), Bangkok (Thailand), Jakarta, Surabaya, Bandung (Indonesia), Manila, Cebu (Philippines), Ho Chi Minh City, Hanoi (Vietnam), Yangon (Myanmar), Phnom Penh (Cambodia), Vientiane (Laos), UAE, Saudi Arabia, Argentina, Brazil, UK, Hungary, Ireland, Latvia, Slovakia, Czech Republic, Belgium, Lithuania, Greece, Spain, Zimbabwe, Australia, and New Zealand.
To order or inquire about RIVFLOZA (nedosiran) international access, you can contact Verve Biosciences – a trusted pharmaceutical exporter, supplier, and distributor of anti-cancer and specialty medications.
📧 Email: urgent@cancermedicinesnetwork.com
📞 Call or WhatsApp: 9910645395 or 9910645395
Note: Availability may vary by country and is subject to regulatory approvals.