Description
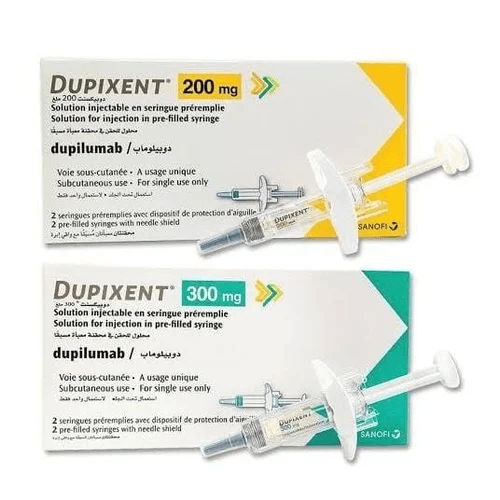
Dupilumab, marketed under the brand name DUPIXENT, It is an prescription medication that has been approved Dupixent (dupilumab) on March 28, 2017 for the treatment of moderate-to-severe atopic dermatitis in adults.
What is DUPIXENT (dupilumab) ?
Dupilumab is indicated for the treatment of moderate-to-severe atopic dermatitis, moderate-to-severe asthma, and for the treatment of chronic rhinosinusitis with nasal polyps (CRSwNP). It has been shown to be effective at treating aspirin-exacerbated respiratory disease (AERD), a typically difficult to treat condition where aspirin intolerant patients have both CRSwNP and asthma.
In May 2022, the indication for dupilumab was updated to include the treatment of eosinophilic esophagitis in people aged twelve years of age and older weighing at least 40 kilograms (88 lb).
In September 2022, the indication for dupilumab was updated to include the treatment of adults with prurigo nodularis (PN).
In March 2023, the EMA approved dupilumab for the treatment of severe atopic dermatitis in children aged six months to five years who are candidates for systemic therapy.
How to order approved “DUPIXENT (dupilumab) for injection” Medicine?
DUPIXENT (Dupilumab) Injection is a prescription medicine, it can be accessed through the Named Patient Program (NPP) for treatment in the following city of India – Bengaluru, Delhi, Chennai, Hyderabad, Mumbai, Pune, Kolkata, Ahmedabad, Gurgaon. To inquire about the cost price of purchasing DUPIXENT and arranging delivery to your location, please get in touch with Mr. Rakesh at +91 9910645395 or send an enquiry to info@cancermedicinesnetwork. We assure you of the quality of the product and guarantee delivery in accordance with your specific requirements.