Description
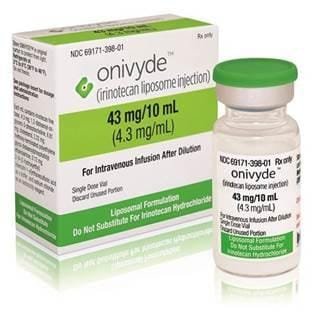
Irinotecan liposome, marketed under the brand name ONIVYDE, It is an prescription medication that has been approved in most major markets including the U.S., Europe and Asia in combination with fluorouracil (FU) and leucovorin (LV) for the treatment of adult patients with metastatic pancreatic adenocarcinoma after disease progression following gemcitabine-base therapy. U.S. Approval: 1996
What is ONIVYDE (irinotecan liposome) ?
ONIVYDE (irinotecan liposome injection), for intravenous use. U.S. Approval: 1996
ONIVYDE is a topoisomerase inhibitor indicated, in combination with fluorouracil and leucovorin, for the treatment of patients with metastatic adenocarcinoma of the pancreas after disease progression following gemcitabine-based therapy. Limitation of Use: ONIVYDE is not indicated as a single agent for the treatment of patients with metastatic adenocarcinoma of the pancreas.
Content Credit: Prescribing Information
Irinotecan liposome injection (PEP02, MM-398) is indicated in combination with fluorouracil and leucovorin for the treatment of patients with metastatic adenocarcinoma of the pancreas that has progressed following gemcitabine-based therapy.
How to order approved “ONIVYDE (irinotecan liposome) for Injection” Medicine?
ONIVYDE (Irinotecan liposome) Injection is a prescription medicine, it can be accessed through the Named Patient Program (NPP) for treatment in the following city of India – Bengaluru, Delhi, Chennai, Hyderabad, Mumbai, Pune, Kolkata, Ahmedabad, Gurgaon. To inquire about the cost price of purchasing ONIVYDE and arranging delivery to your location, please get in touch with Mr. Rakesh at +91 9910645395 or send an enquiry to info@cancermedicinesnetwork. We assure you of the quality of the product and guarantee delivery in accordance with your specific requirements.