Description
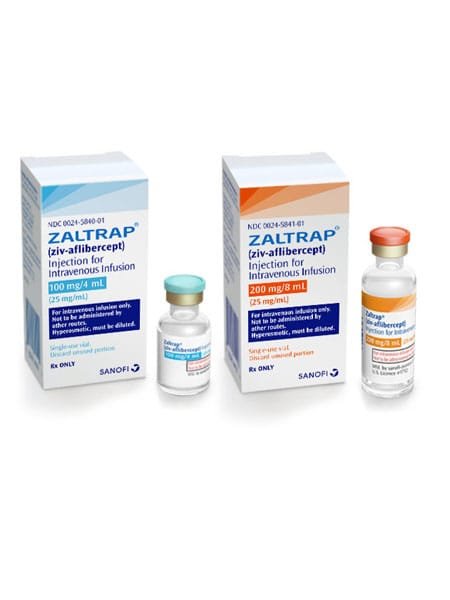
Ziv-Aflibercept, marketed under the brand name ZALTRAP, It is an prescription medication that has been approved Ziv-Aflibercept, also known by the brand name Zaltrap, on August 3, 2012. The European Commission approved Zaltrap for marketing in the European Union on February 1, 2013.
What is Zaltrap (Ziv-aflibercept) ?
ZALTRAP (ziv-aflibercept) Injection for Intravenous Infusion. Initial U.S. Approval: 2012
Zaltrap is used in combination with other medications to treat colorectal cancer. Zaltrap is usually given after other medications have been tried without successful treatment.
Aflibercept is a biopharmaceutical drug invented by Regeneron Pharmaceuticals, approved in the United States and Europe for the treatment of wet macular degeneration under the trade name Eylea, and for metastatic colorectal cancer as Zaltrap.
How to order approved “Zaltrap (Ziv-aflibercept) for Injection” Medicine?
ZALTRAP (Ziv-Aflibercept) Injection is a prescription medicine, it can be accessed through the Named Patient Program (NPP) for treatment in the following city of India – Bengaluru, Delhi, Chennai, Hyderabad, Mumbai, Pune, Kolkata, Ahmedabad, Gurgaon. To inquire about the cost price of purchasing ZALTRAP and arranging delivery to your location, please get in touch with Mr. Rakesh at +91 9910645395 or send an enquiry to info@cancermedicinesnetwork. We assure you of the quality of the product and guarantee delivery in accordance with your specific requirements.